PPT Ch. 9 and 10 Chemical Bonding PowerPoint Presentation, free
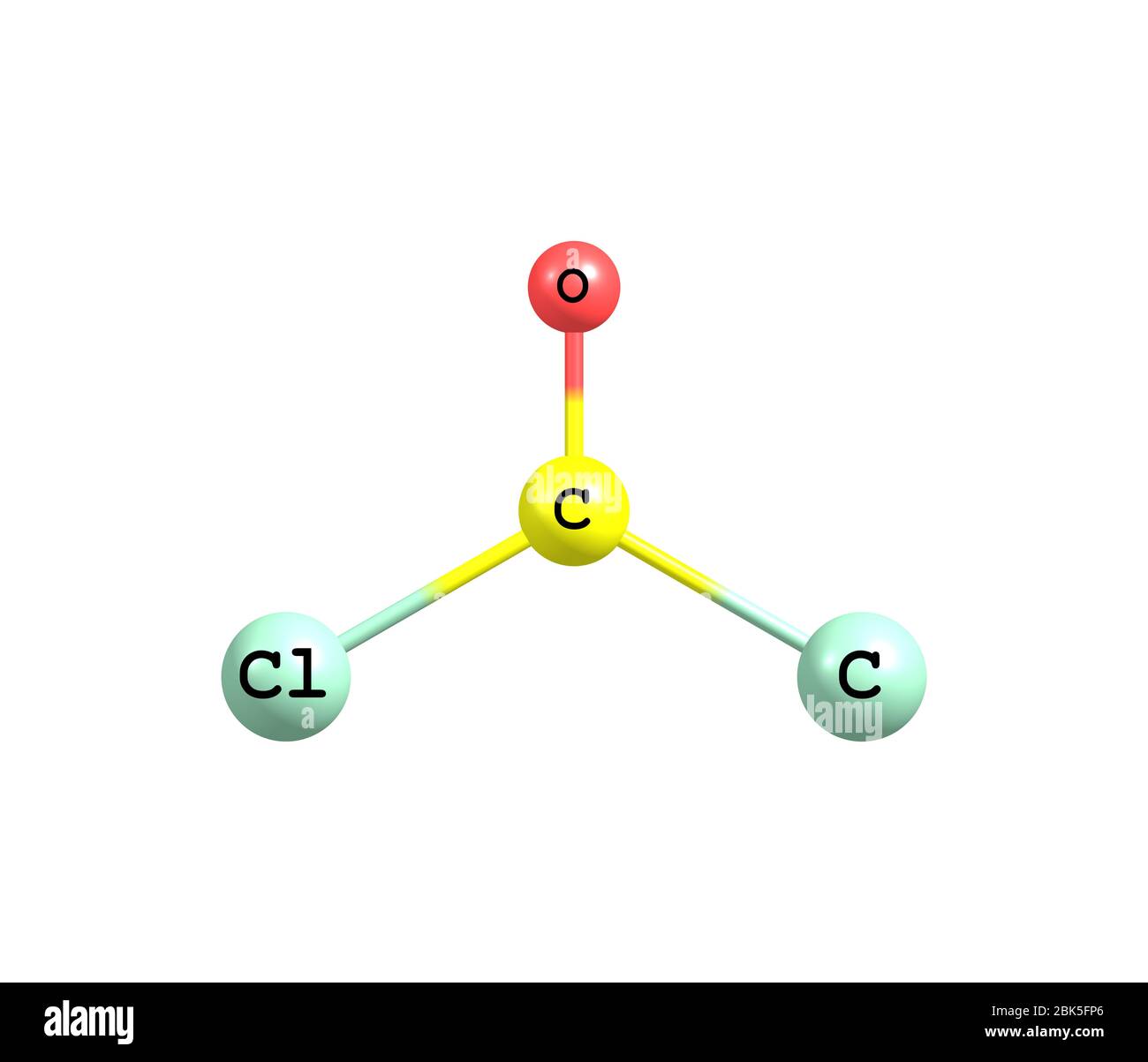
is the chemical compound with the formula COCl2. This colorless gas
It is an inorganic compound that comprises Cobalt and Chlorine atoms. CoCl2 is a crystalline solid that is sky-blue in color. It is readily soluble in water, alcohol, and acetone. It occurs at different levels of hydration as dehydrates and hexahydrates. These versions of the salt are purple and pink, respectively.
PPT The Shapes of Molecules PowerPoint Presentation, free download
What is this molecule? COCl 2 is the chemical formula for carbonyl chloride, also known as phosgene. It is a colorless gas at room temperature with a pungent odor. Phosgene is a highly toxic compound that was historically used as a chemical warfare agent during World War I.
The Shapes of Molecules презентация онлайн
Hi Guys!COCl2 is a chemical formula for a Phosgene molecule. It comprises one Carbon, one Oxygen, and two Chlorine atoms. In this video, we share a step-by-s.
COCl2 Phosgene
Molecular Formula Cl Co Average mass 129.839 Da Monoisotopic mass 128.870911 Da ChemSpider ID 22708 - Charge More details: Names Properties Searches Spectra Vendors Articles More Names and Synonyms Database ID (s) Validated by Experts, Validated by Users, Non-Validated, Removed by Users Cobalt (II) chloride [Wiki] 231-589-4 [EINECS] 7646-79-9 [RN]
18+ Electron Geometry Vs Molecular Geometry Gif GM
By Aparna Dev The Lewis structure of COCl2, also known as carbonyl chloride or phosgene, is a representation of its molecular structure using Lewis symbols and lines to show the bonding between atoms. In this structure, carbon is the central atom bonded to two oxygen atoms and one chlorine atom.

Draw the Lewis structure for COCl2, including lone pairs.What is the
In the Lewis structure for COCl 2 there are a total of 24 valence electrons. You'll need to form a double bond between the Carbon and Oxygen to complete the octet on the Carbon atom. Transcript: Let's do the Lewis structure for COCl2. Carbon, in group 4 or 14, has 4 valence electrons. Oxygen in group 6, sometimes called 16, 6 valence electrons.

Draw the Lewis structure for COCl2, including lone pairsWhat is the
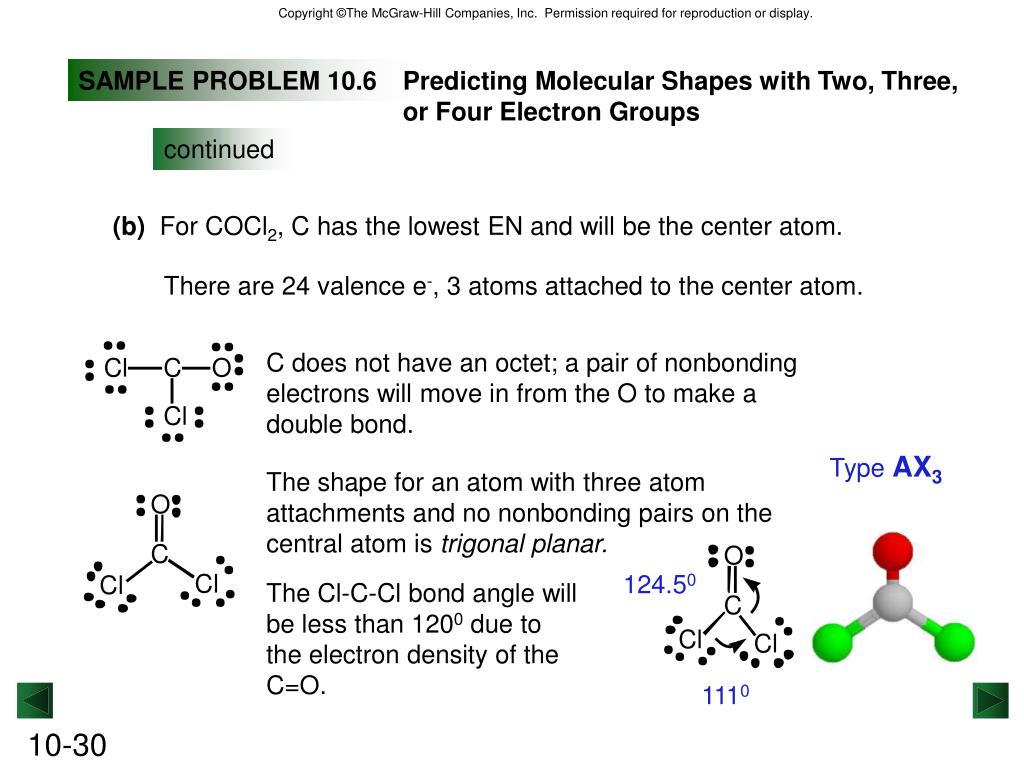
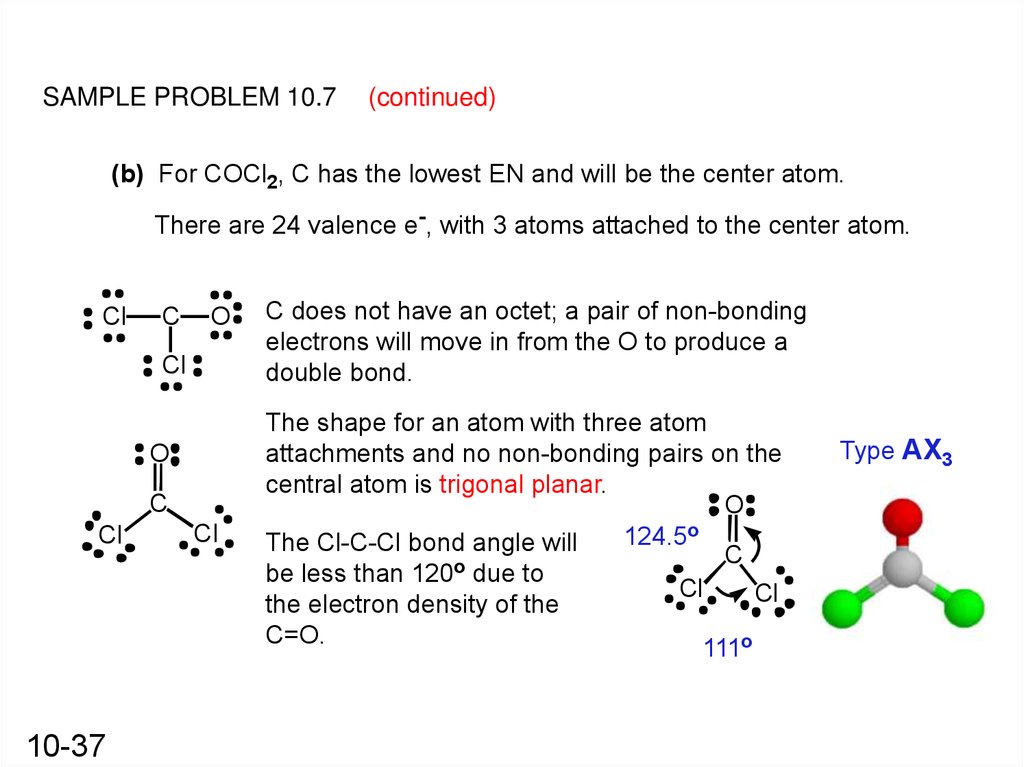
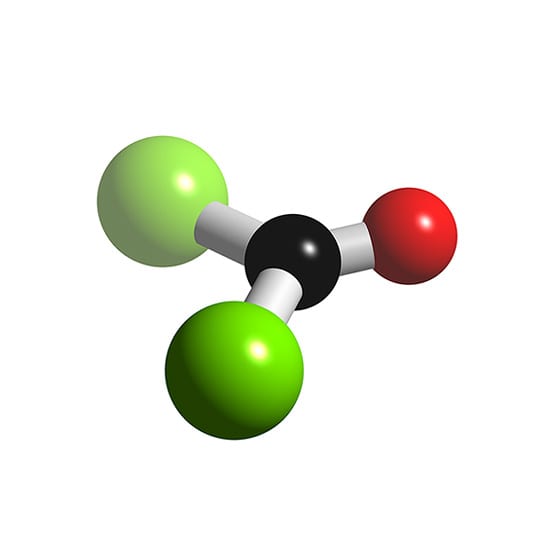
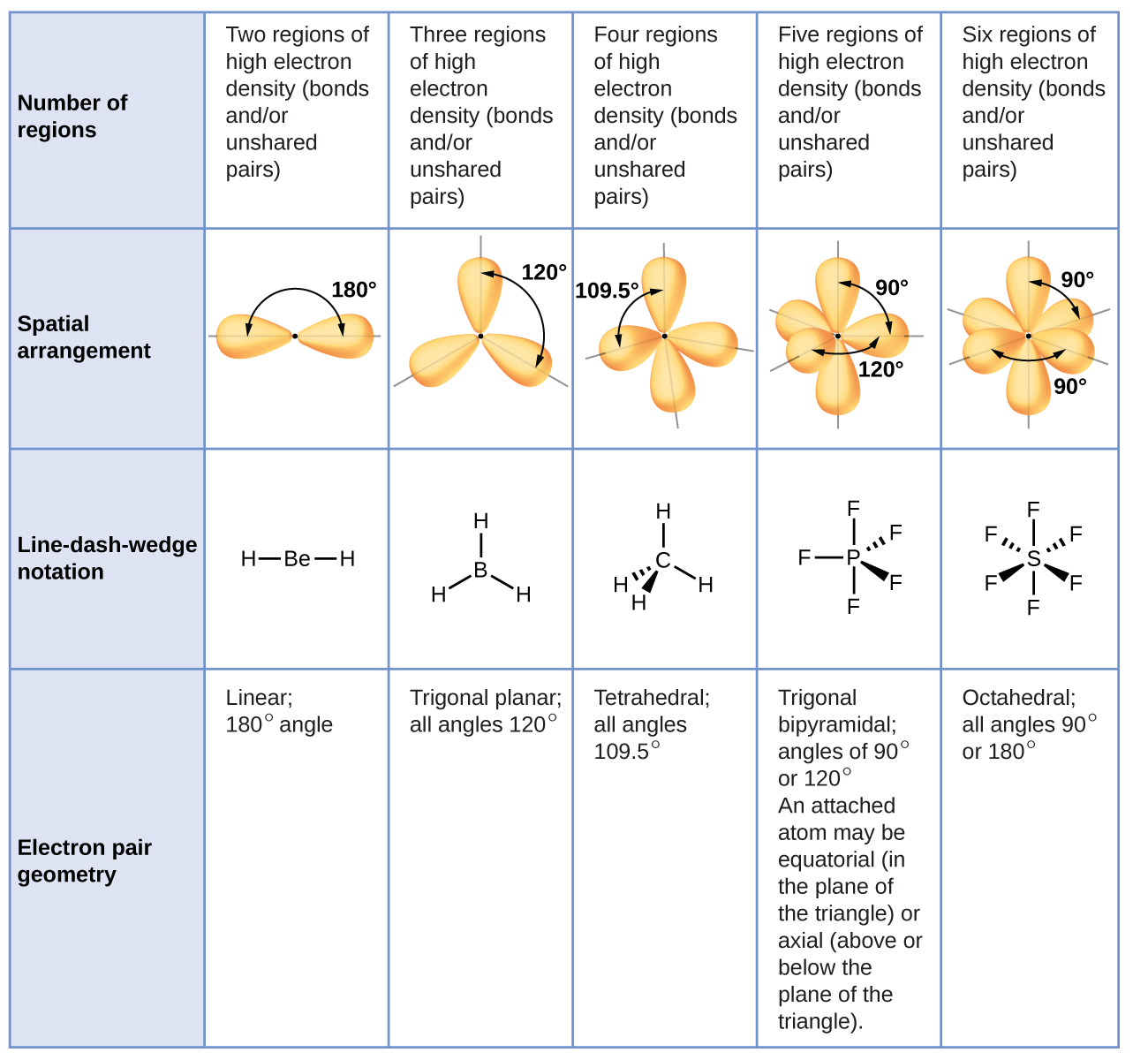
The molecular shape of COCl 2 is trigonal planar, or AX 3 using Valence Shell Electron Pair Repulsion (VSEPR) theory. Hence, the molecular geometry of COCl 2 only has 120 degree bond angles in the molecule. COCl 2 looks like this: How do you find the molecular geometry of COCl2?
[Solved] what is the lewis structure, electron geometry, molecular
Molecular Formula CCl2O COCl2 Synonyms PHOSGENE Carbonyl dichloride Carbonic dichloride Carbonyl chloride Phosgen View More. Molecular Weight 98.91 g/mol Computed by PubChem 2.2 (PubChem release 2021.10.14) Dates Create: 2004-09-16 Modify: 2023-12-30 Description Phosgene is a colorless nonflammable gas that has the odor of freshly cut hay.

COCl2 Lewis Structure How to Draw the Lewis Structure for COCl2 YouTube
Molecular Formula Cl2Co Synonyms 7646-79-9 Cobaltchloride Cobalt chloride (CoCl2) MFCD00010938 Cobalt (II) chloride, ultra dry View More. Molecular Weight 129.84 g/mol Computed by PubChem 2.2 (PubChem release 2021.10.14) Dates Create: 2005-03-26 Modify: 2023-12-30 Description Formerly approved as a food additive but now prohibited

a Structural descriptions of an optimized free phosgene (COCl2
The IUPAC name for the COCl 2 molecule is carbonyl dichloride. It is a toxic, colorless gas that emits a suffocating odor. It is more commonly known as phosgene. Phosgene is particularly important as a reagent in the polymer and plastic manufacturing industry.

COCl2 Molecular Geometry, Bond Angles (Phosgene) YouTube
The crystal unit of the solid hexahydrate CoCl 2 •6 H 2O contains the neutral molecule trans - CoCl 2(H 2O) 4 and two molecules of water of crystallization. [8] This species dissolves readily in water and alcohol . The anhydrous salt is hygroscopic and the hexahydrate is deliquescent. [citation needed]
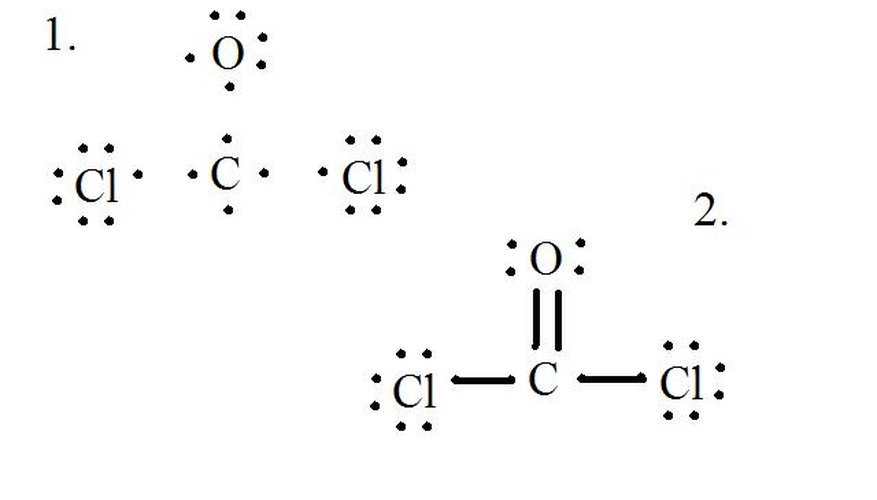
How to Calculate the Formal Charge of CoCl2 Sciencing
In the COCl 2 Lewis structure, there are two single bonds and one double bond around the carbon atom, with two chlorine atoms and one oxygen atom attached to it. Two chlorine atoms with single bonds have three lone pairs, and one oxygen atom with a single bond has two lone pairs. COCl2 Lewis Structure - How to Draw the Lewis Structure for COCl2
Best Cocl2 Lewis Structure Molecular Geometry Pics GM
3a. Drawing the structure of CoCl2 Table view List view 3a. Drawing the structure of CoCl2 COCI Lone pairs of electrons (central atom) Bonding groups (central atom) Total valence electrons VSEPR Molecular shape (central atom) Choose Choose. 3b. Evaluating the structure of CoCl2 Table view List view 3b.
Seite nicht gefunden telekine fernsehproduktion
What is the molecular shape of COCl2? The C - Cl bond in COCl2 is polar or nonpolar? What is the Cl - C - Cl bond angle? The molecule COCl2 is polar or nonpolar? This problem has been solved! You'll get a detailed solution from a subject matter expert that helps you learn core concepts. See Answer

COCl2 Lewis Structure, Molecular Geometry, Hybridization, and Polarity
Structure and basic properties Phosgene is a planar molecule as predicted by VSEPR theory. The C=O distance is 1.18 Å, the C−Cl distance is 1.74 Å and the Cl−C−Cl angle is 111.8°. [9] Phosgene is a carbon oxohalide and it can be considered one of the simplest acyl chlorides, being formally derived from carbonic acid . Production

COCl2 (Phosgene) Molecular Geometry, Bond Angles (and Electron Geometry
COCl2 molecule consists of one C, one O, and Cl atoms. The total number of valence electrons = 4 + 6 + 7*2 = 10 + 14 = 24. Step 2: Now, we will have to find out the element which will take up the position of the central atom.