Lewis Dot Diagram For H2o Free Diagram For Student

Lewis Structure of Water H2O YouTube
---- Steps to Write Lewis Structure for compounds like H2O ---- 1. Find the total valence electrons for the H2O molecule. 2. Put the least electronegative atom in the center. Note: Hydrogen.
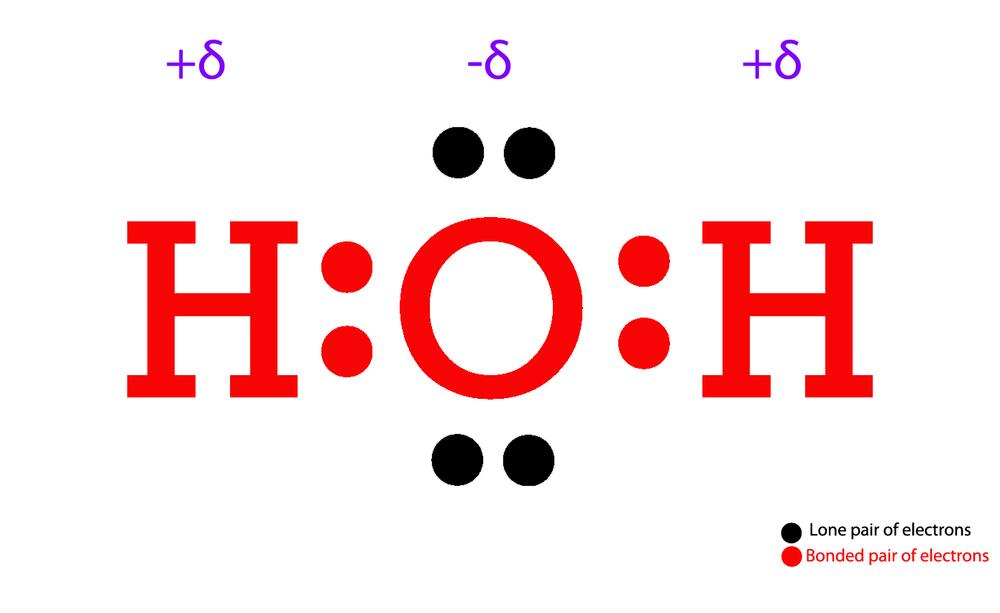
Draw Step By Step The Lewis Structure For Water (H2O)
Draw a single bond between each hydrogen atom and the oxygen atom to account for 4 valence electrons. Place the remaining 4 valence electrons around the oxygen atom as lone pairs. Check to see if each atom has a full octet of electrons (except for hydrogen, which only needs 2 electrons).

【4 Steps】H2O Lewis StructureLewis Structure for H2O (Water)Lewis Dot Structure for Water(H2O)
Step #1: Calculate the total number of valence electrons. Here, the given molecule is H2O (water). In order to draw the lewis structure of H2O, first of all you have to find the total number of valence electrons present in the H2O molecule. (Valence electrons are the number of electrons present in the outermost shell of an atom).

H2O Lewis Structure ,Valence Electrons ,Formal Charge,Polar or Nonpolar
We can illustrate the formation of a water molecule from two hydrogen atoms and an oxygen atom using Lewis dot symbols: The structure on the right is the Lewis electron structure, or Lewis structure, for H 2 O. With two bonding pairs and two lone pairs, the oxygen atom has now completed its octet. Moreover, by sharing a bonding pair with oxygen.

H2O Lewis structure and Molecular Geometry [No1 Best Explanation] Science Education and Tutorials
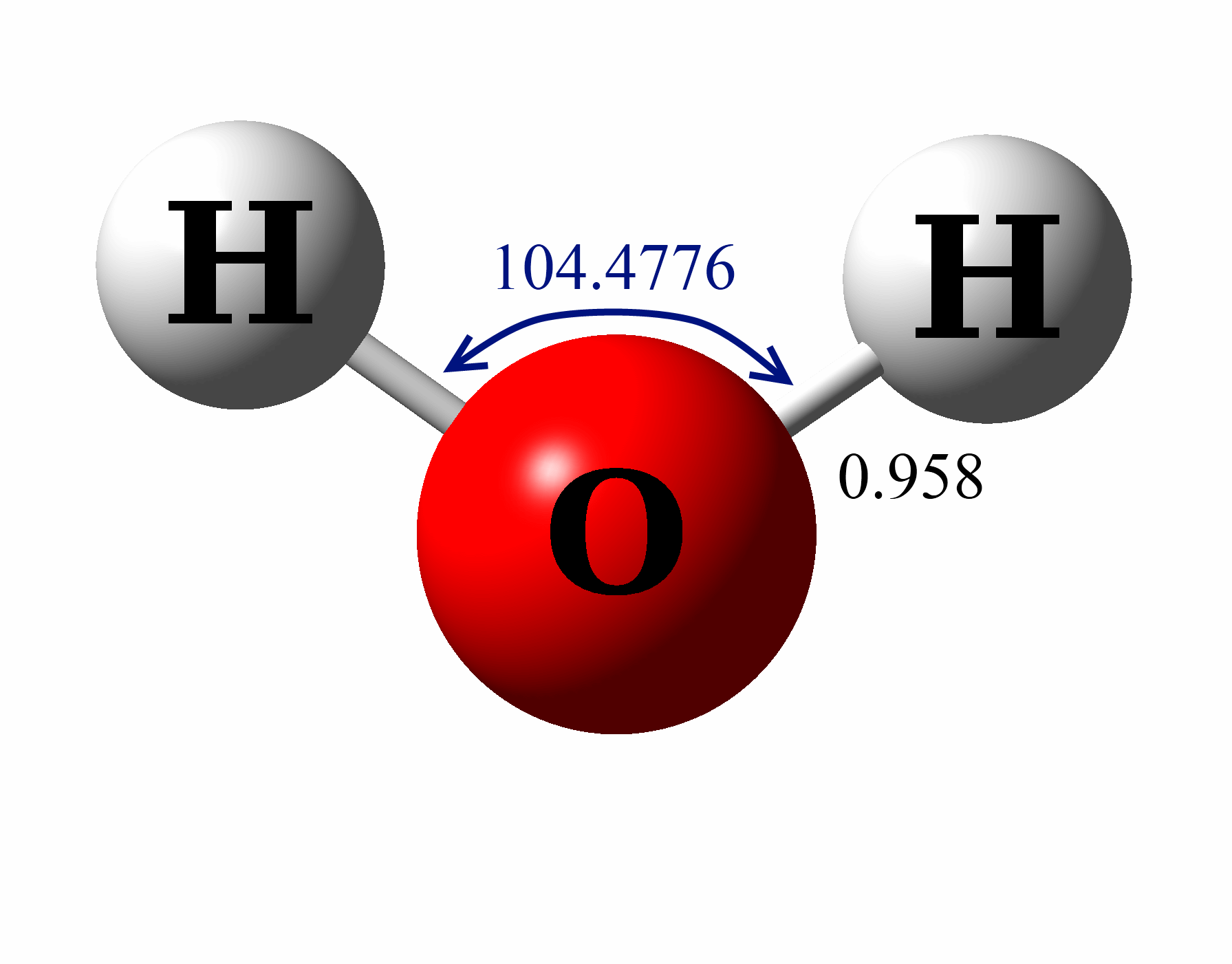
Lewis Structure of H 2 O indicating bond angle and bond length Water ( H 2O) is a simple triatomic bent molecule with C 2v molecular symmetry and bond angle of 104.5° between the central oxygen atom and the hydrogen atoms.

H2O Lewis Structure, Molecular Geometry, and Hybridization Techiescientist
Shop the formula made with a patented protein blend that gets closest to breast milk. Made with certified-clean ingredients and clinically proven benefits. Shop ByHeart now!

Lewis structures StudyPug
A Lewis structure is a way to show how atoms share electrons when they form a molecule. Lewis structures show all of the valence electrons in an atom or molecule.. In a water molecule, an oxygen atom forms two bonds, one to each hydrogen atom. Chemists normally represent a bond using a line instead of two dots. The structures of H 2,.
Lewis Dot Diagram For H2o Free Diagram For Student
H = 1 - ½ (2) = 0; O = 6 - 4 - ½ (4) = 0. The Lewis structure of H₂O is. Answer link. You can find a procedure for drawing Lewis structures at this location. For H₂O, O must be the central atom The skeleton structure is H-O-H. O has 6 valence electrons, and each H has one. You must arrange 8 electrons in pairs so that O has 8 and each H.

H2O Lewis structure and Molecular Geometry [No1 Best Explanation] Science Education and Tutorials
The Lewis structure of hydrogen and 2 oxygen atoms shows a total of eight valence electrons participate in the bond formation to form a single triatomic H2O molecule. Here, we need to understand how the Lewis structure is drawn for the H2O molecule: Look for the total valence electrons: It is eight to form a single H2O molecule.

H2O Lewis Structure Lewis Dot Structure for H2O Lewis Structure of H2OWater Lewis
💡 Tutoring, publications, website, reading notes, guides: https://linktr.ee/liahtutoring⛵Contact: [email protected] video explains how to find the.

H2O Lewis structure and Molecular Geometry [No1 Best Explanation] Science Education and Tutorials
Valence electrons of Hydrogen: 1*2 ( as there are 2 Hydrogen atoms, we will multiply it by 2) Valence electrons of Oxygen: 6 Total number of valence electrons in H2O: 2 + 6 = 8 valence electrons Thus, H2O has a total of 8 valence electrons. H2O Lewis Structure
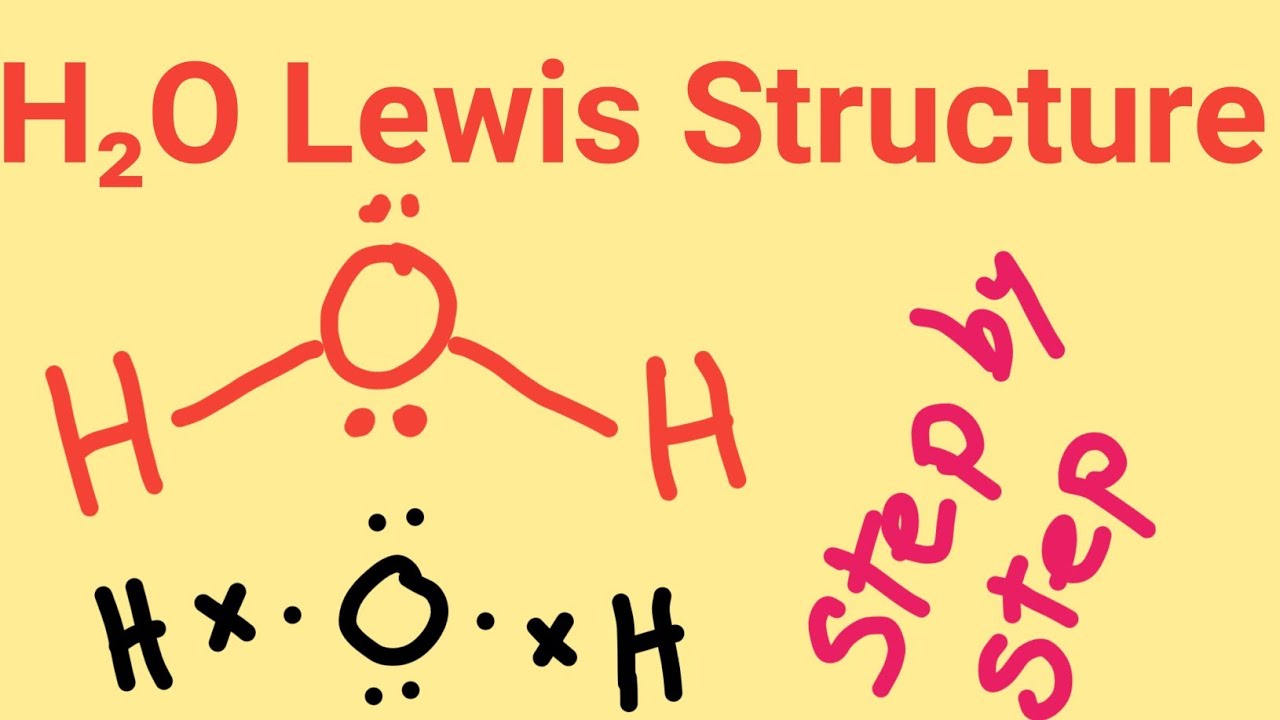
H2O Lewis StructureHow to draw H2O Lewis Structure (Water)H2O Electron Dot Structure for
A step-by-step explanation of how to draw the H2O Lewis Dot Structure (Water).For the H2O structure use the periodic table to find the total number of valenc.

H2O Lewis Structure, Molecular Geometry, and Hybridization Techiescientist
We can illustrate the formation of a water molecule from two hydrogen atoms and an oxygen atom using Lewis dot symbols: The structure on the right is the Lewis electron structure, or Lewis structure, for H 2 O. With two bonding pairs and two lone pairs, the oxygen atom has now completed its octet. Moreover, by sharing a bonding pair with oxygen.

【4 Steps】H2O Lewis StructureLewis Structure for H2O (Water)Lewis Dot Structure for Water(H2O)
Figure 5.2.9 5.2. 9: (a) H2O has four regions of electron density around the central atom, so it has a tetrahedral electron-pair geometry. (b) Two of the electron regions are lone pairs, so the molecular structure is bent. Exercise 5.2.3 5.2. 3. The hydronium ion, H 3 O +, forms when acids are dissolved in water.

In this video we are going to learn about the Lewis structure of H2O. It is a chemical formula
The Lewis structure, also known as an electron dot structure, is a diagrammatic representation of determining the total number of valence electrons present in an atom that are ready to form bonds to form a molecule and, eventually, a compound. Table of Contents How to draw Lewis Structure for H 2 O Molecular Geometry of H 2 O Hybridization of H 2 O
Gambar Struktur Lewis H2O pulp
The Lewis Structure of H2O molecule reveals that two hydrogen atoms are linked to one oxygen atom. A single covalent link is formed when each hydrogen atom and oxygen atom share one electron. Furthermore, the oxygen atom has two lone pairs of electrons that are not shared by any other element.